Calculate the formal charge on each atom :C⁻≡O⁺:ĩ. As before, add valence electrons to give each atom an octet :C≡O:Ĩ. Draw a new trial structure, this time inserting one double bond for each extra pair of electrons : C≡O.ħ. The trial structure has four extra electrons.Ħ. Now count the valence electrons you actually have available. Count the valence electrons in your trial structure (14).ĥ. In this editor, I will have to write it as :::C-Ö::Ĥ. Draw a trial structure by putting electron pairs around every atom until each gets an octet. Draw a skeleton structure in which the other atoms are single-bonded to the central atom: C-O.ģ. That will normally be the least electronegative atom (C).Ģ. Decide which is the central atom in the structure. 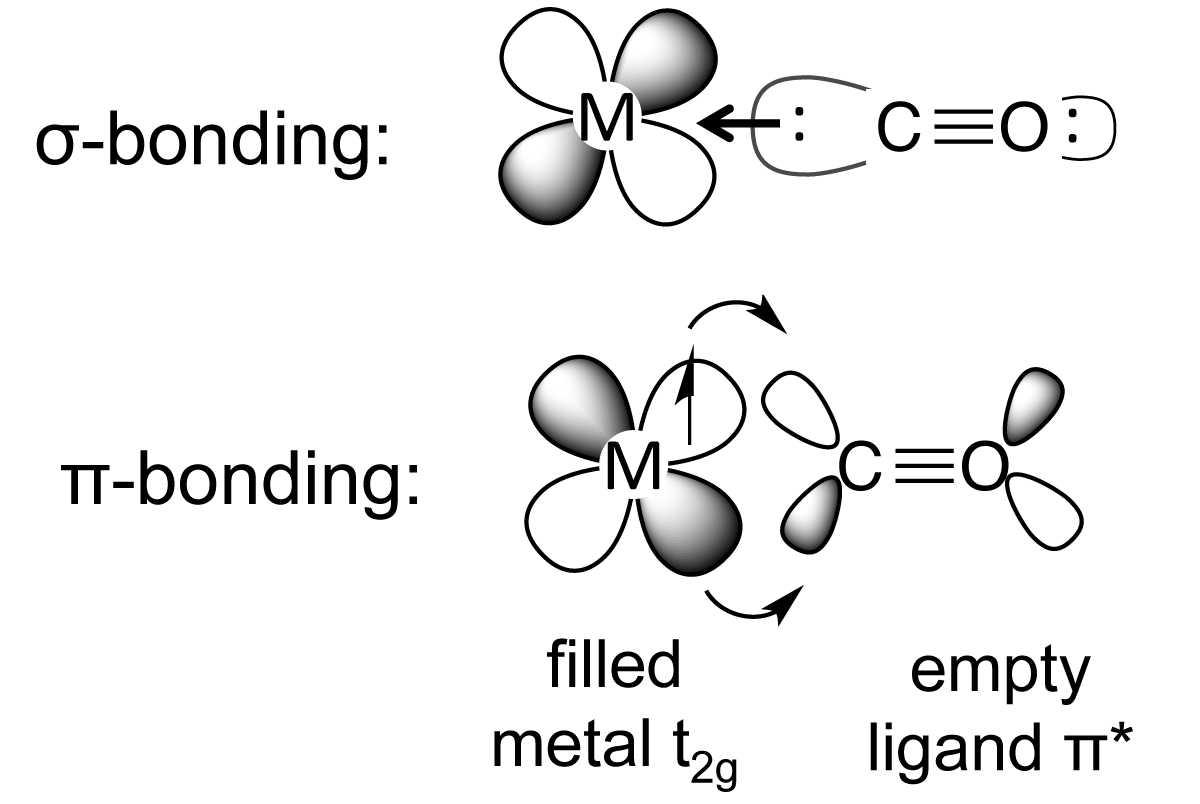
In the reactions below, the nucleophiles water and hydroxide react at the carbon.Here are the steps that I follow when drawing a Lewis structure.ġ. The carbon in CO 2 is electron-poor and it reacts with nucleophiles. In the reactions below, the CO molecule complexes to a nickel center. We have seen that the carbon of CO is electron-rich. Solubility in water (1.013 bar and 0 deg C (32 deg F)) = 1.7163 vol/vol.Boiling point (Sublimation) = -78.5 deg C.It reacts with water to form carbonic acid. It freezes at -78.5 deg C at normal atmospheric pressure to form "dry ice" without going through a liquid phase. CO 2 gas has a slightly irritating odor, is colorless and heavier than air. It is assimilated by plants in photosynthesis. 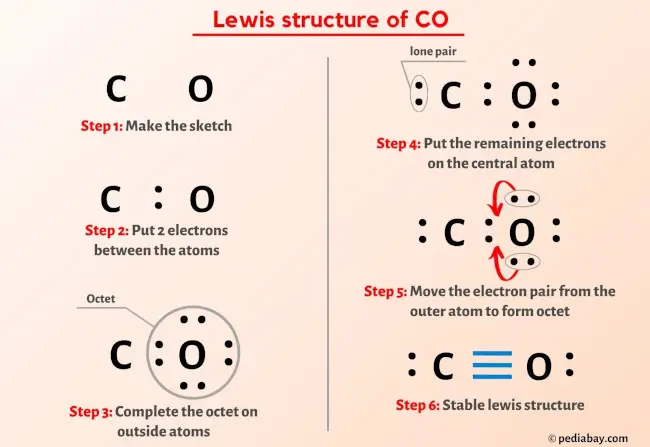
Solubility in water (1.013 bar and 0 deg C (32 deg F)) = 0.0352 vol/volĬarbon dioxide is produced from combustion, fermentation, and is a product of respiration in animals.It binds strongly with hemoglobin, the oxygen-carrying protein in blood and prevents oxygen binding. The ultimate products are CO 2 and H 2O.Ĭarbon monoxide is flammable and it is very toxic and odorless. Soot, C s, is also a product.īelow is a simplified scheme that shows the principle reactions in the combustion of acetylene, C 2H 2, in a flame. Mechanistic studies show multiple steps that product both larger and smaller hydrocarbon and oxyhydrocarbon species before the ultimate formation of water and carbon dioxide. None of these reaction schemes gives a true picture of combustion in a flame. The combustion of acetylene is also exothermic. The enthalpy of formation of any element in its most common form is zero. You can calculate the enthalpy of reaction by subtracting the enthalphies of formation of products from reactants. Below is the balanced reaction for the combustion of isobutane. It is important to note that the sigma and the pi orbitals extend over the whole 3-atom molecule.Ĭombustion of any organic molecule in a minimum of oxygen produces carbon monoxide. 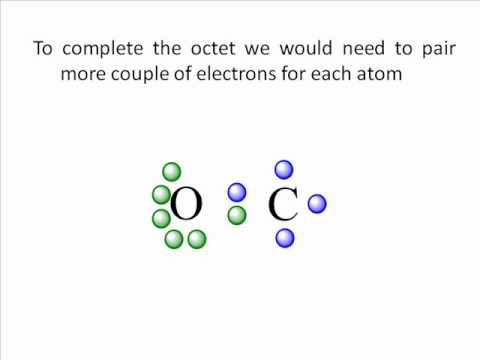
The remaining 4 p orbitals on the oxygen atoms are non-bonding molecular orbitals. This makes 2 pi bonding molecular orbitals and 2 pi antibonding molecular orbitals. The carbon 2p(x) combines with the group orbital O2p(x)+O2p(x) and the carbon 2p(y) combines with the group orbital O2p(y)+O2p(y). In carbon dioxide, the carbon 2s and 2p(z) orbitals combine with 2 combination orbitals (O2s+O2s and O2s-O2s) to make 2 sigma bonding and 2 sigma antibonding orbitals. One of the non-bonding pairs is closer in energy to the carbon atomic orbitals and the other is closer in energy to the oxygen atomic orbitals. There are also two orbitals that, together, are nonbonding. Oxygen is more electronegative than carbon, so its orbitals are more highly stabilized and lower in energy than the carbon orbitals.īoth of these molecules have 3 filled bonding orbitals. Carbon, with 4 valence electrons, and oxygen with 6 valence electrons, together have the same number of electrons as dinitrogen. The molecular orbital diagram of carbon monoxide is very similar to that of molecular nitrogen. Carbon dioxide is electron-poor at the central carbon and acts as an electrophile. The Lewis structure of carbon monoxide shows that it is relatively electron-rich at carbon. There is a C-O triple bond in CO with formal charges on each atom.Ĭarbon dioxide has a formal double bond between C-O. The carbon oxides are stable, diamagnetic molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
